What is Deep Brain Stimulation (DBS)?
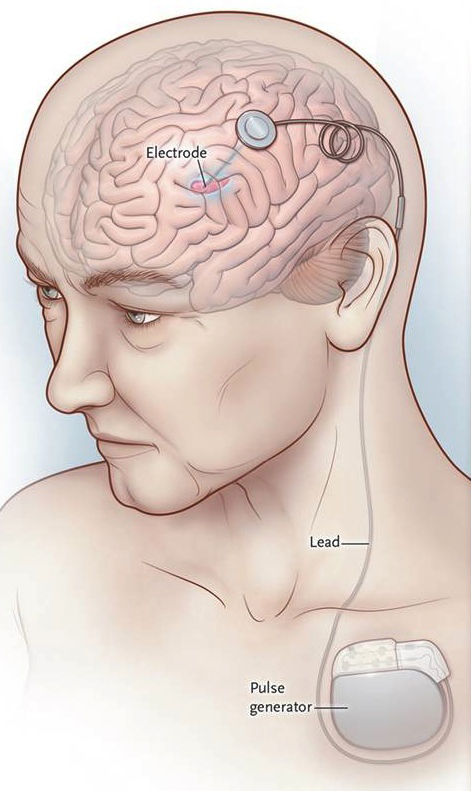
DBS is a treatment used to provide relief to individuals experiencing painful muscle movements caused by dystonia (involuntary muscle spasms) and/or spasticity (stiff or rigid muscles).
DBS involves surgically placing electrodes in the brain, attaching the electrodes to wires that lead to battery-operated neurostimulator (pulse generator) implanted in the chest.
The neurostimulator sends electrical pulses to certain parts of the brain to shut them down and stop them from sending the signals that cause painful dystonia or spasticity.
How effective is it?
- DBS has shown good short-term results in NBIA
- However, the benefits of DBS seem to decrease over time
When should it be considered?
- If oral baclofen isn’t effective anymore, individuals can consider moving to DBS (or a baclofen pump)
- Certain doctors may choose DBS over a baclofen pump if they have more experience with DBS and/or have a DBS clinic nearby for referral
- It is rare for an individual to have both DBS and a baclofen pump, but does sometimes happen
Are there any side effects or cons?
The benefits of DBS seem to decrease over time in NBIA.
Side-effects are rare in centers/hospitals with DBS experience.
- The most severe risk is possible bleeding in the brain
- There may be some surgery risks:
- Infections
- Seizures
- Allergic reaction to implanted materials
- Other side effects of DBS surgery include:
- Increased dystonia (involuntarily muscle contraction and spasms)
- Dysarthria (poor articulation or slurring of speech)
- Dysphasia (difficulty in using or understanding words)
- These side effects are usually reversible
The risks associated with DBS are considered acceptable if an individual is expected to benefit from the procedure.
There is a limited amount of information available to help guide the setting of the stimulator in NBIA patients. This process can involve some trial and error. It is helpful to work with a physician experienced in adjusting the settings.
Copyright © 2014 by NBIAcure.org. All rights reserved.