If you or a loved one has recently been diagnosed with CoPAN, we hope our website can help answer some of your questions and guide you to the most appropriate resources. We encourage you to call our team to speak to a genetic counselor who can help you navigate this diagnosis. Please email us at info@nbiacure.org and we will set up a phone call with you.
CoPAN (COASY Protein-Associated Neurodegeneration) is an NBIA disorder that is characterized by spaticity and weakness of the lower limbs early in the disease and iron accumulation and calcifications found in the globus pallidus.
SYMPTOMS
Symptoms of CoPAN typically start to appear in childhood. There are only a few cases of CoPAN that have been reported, so information about CoPAN may change once more affected individuals are identified.
Common symptoms include:
Spastic-dystonic paraparesis
- Lower limbs spasticity (stiff or rigid muscles), dystonia (involuntary muscle contraction and spasms), and weakness
- Develops early in the disease
Oro-mandibular dystonia
- Involuntarily muscle contraction and spasms of the mouth, jaw and tongue
- Develops later in the disease
Dysarthria (speech problems)
- Difficulty using or controlling the muscles of the mouth, tongue, larynx or vocal cords which are used to make speech
- This can make a person’s speech difficult to understand in several different ways, including stuttering, slurring, or soft or raspy speech
Axonal neuropathy
- Nerve cells begin to function abnormally because the axons (which send signals from one nerve cell to another) are deteriorating
Parkinsonism (symptoms similar to Parkinson’s disease)
- Bradykinesia (slow movement)
- Rigidity (stiffness)
- Tremors (shaking)
Cognitive impairment
- More research is needed to determine the level of impairment
Obsessive-compulsive behavior
CAUSE/GENETICS
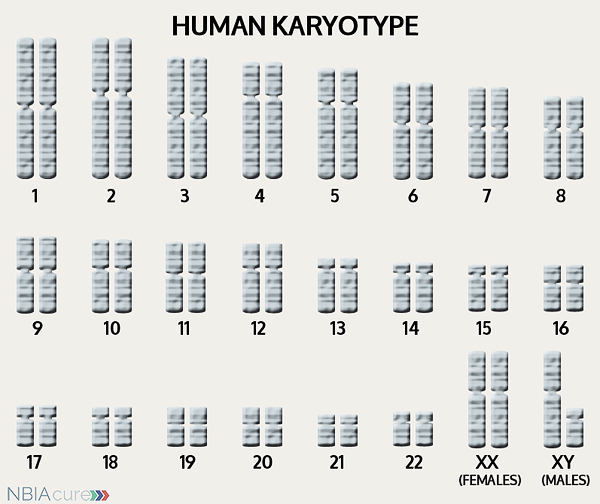 The human body is made up of millions of cells. Inside every cell there is a structure called DNA, which is like an instruction book. DNA contains detailed steps about how all the parts of the body are put together and how they work. However, DNA contains too much information to fit into a single “book,” so it is packaged into multiple volumes called chromosomes. Humans typically have 46 total chromosomes that are organized in 23 pairs. There are two copies of each chromosome because we receive one set of 23 chromosomes from our biological mother and the other set of 23 from our biological father. Chromosomes 1-22 are called autosomes and the last pair is called the sex chromosomes because they determine a person’s gender. Females have two X chromosomes and males have one X and one Y.
The human body is made up of millions of cells. Inside every cell there is a structure called DNA, which is like an instruction book. DNA contains detailed steps about how all the parts of the body are put together and how they work. However, DNA contains too much information to fit into a single “book,” so it is packaged into multiple volumes called chromosomes. Humans typically have 46 total chromosomes that are organized in 23 pairs. There are two copies of each chromosome because we receive one set of 23 chromosomes from our biological mother and the other set of 23 from our biological father. Chromosomes 1-22 are called autosomes and the last pair is called the sex chromosomes because they determine a person’s gender. Females have two X chromosomes and males have one X and one Y.
If DNA is the body’s instruction book and it is stored in multiple volumes (called chromosomes), then genes would be the individual chapters of those books. Genes are small pieces of DNA that regulate certain parts or functions of the body. Sometimes multiple genes (or chapters) are needed to control one function. Other times, just one gene (or chapter) can influence multiple functions. Since there are two copies of each chromosome, there are also two copies of each gene. In some gene pairs, both copies need to be expressed (or turned on) in order for them to do their job correctly. For other genes pairs, only one copy needs to be expressed.
When a single cell in the human body divides and replicates, its DNA is also replicated. This replication process is usually very accurate but sometimes the body can make a mistake and create a “typo” (or mutation). Just like a typo in a book, a mutation in the DNA can be unnoticeable, harmless, or serious. A mutation with serious consequences can result in a part of the body not developing correctly or a particular function not working properly.
In the case of NBIA disorders, changes in certain genes cause a person to develop their particular type of NBIA. Changes in these NBIA genes lead to the groups of symptoms we observe, although we do not yet understand how the changed genes cause many of these findings. COASY is the only gene known to cause CoPAN. COASY’s main job is to tell the body’s cells how to make an enzyme called coenzyme A synthase. It is not yet clear to us how a decrease in this enzyme eventually leads to iron accumulation in the brain
As mentioned earlier, humans have a total of 23 pairs of chromosomes. Half of these chromosomes are passed down (or inherited) from the biological mother and half from the biological father. The way in which a gene change is passed down from parents to child varies from gene to gene. The COASY gene that is altered in those with CoPAN is inherited in an autosomal recessive manner.
“Autosomal” refers to the fact that the COASY gene is located on chromosome 17, which is one of the autosomes (chromosome pairs 1-22). Since the sex chromosomes are not involved, males and females are equally likely to inherit a mutation located in the autosomes. “Recessive” refers to the fact that a mutation must be present in both copies of the COASY gene for a person to have CoPAN. If an individual has only one COASY mutation, then they are called a “carrier” for CoPAN. Carriers do not have health problems related to the mutation and often do not know they carry a recessive gene mutation. However, if two CoPAN carriers have a child together, then there is a 25% chance that they will both pass on their recessive COASY gene mutations and have a child with CoPAN.
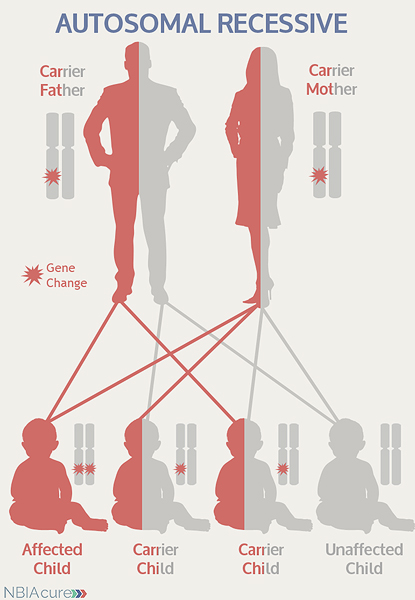 As seen in the image to the left, in a pregnancy between two CoPAN carriers:
As seen in the image to the left, in a pregnancy between two CoPAN carriers:
- There is a 25% chance of the child having CoPAN
- There is a 50% chance that the child will be a carrier like his/her parents
- There is a 25% chance that the child will not have CoPAN or be a carrier
DIAGNOSIS & TESTING
A brain MRI is a standard diagnostic tool for all NBIA disorders. MRI stands for magnetic resonance imaging. An MRI produces a picture of the body that is created using a magnetic field and a computer. The technology used in an MRI is different from that of an x-ray. An MRI is painless and is even considered safe to do during pregnancy. Sometimes an MRI is done of the whole body, but more often, a doctor will order an MRI of one particular part of the body.
Evidence of iron accumulation on a brain MRI is often an important clue leading to the diagnosis of CoPAN. A T2 sequence is the preferred type of MRI for NBIA diagnosis because it is highly sensitive to the detection of brain iron.
MRI findings for CoPAN include:
- Hypointensity (darkness) in the substantia nigra and globus pallidus on T2 MRI
- The dark patches indicate iron accumulation
- Appear as a central region of hyperinternsity (brightness) in the middle of a dark patch of iron accumulation in the globus pallidus and resemble the eye-of-the-tiger sign seen in the PKAN type of NBIA
- However, additional testing using an axial CT showed that the bright areas were calcifications
Diagnosis of CoPAN can be confirmed through genetic testing of the COASY gene to find gene changes. Genetic testing is done through sequence analysis.
Rarely, an individual with the signs and symptoms of CoPAN may have only one or no COASY gene changes identified. This can happen because the genetic testing is not perfect and has certain limitations. It does not mean the person does not have CoPAN; it may just mean we do not yet have the technology to find the hidden gene changes. In these cases it becomes very important to have doctors experienced with CoPAN review the MRI and the person’s symptoms very carefully to be as sure as possible of the diagnosis.
MANAGEMENT
 There is no standard treatment for CoPAN. Diagnosed individuals are managed by a team of medical professionals that recommends treatments based on current symptoms.
There is no standard treatment for CoPAN. Diagnosed individuals are managed by a team of medical professionals that recommends treatments based on current symptoms.
After diagnosis, individuals with CoPAN are recommended to get the following evaluations to determine the extent of their disease:
- Neurologic examination for dystonia, rigidity, and spasticity, and parkinsonism
- Evaluation of ambulation and speech
- Developmental assessment
- Assessment for physical therapy, occupational therapy, and/or speech therapy
- Psychiatric assessment for possible obsessive-compulsive behavior
- Medical genetics consultation
Dystonia (involuntarily muscle contraction and spasms) can be debilitating and distressing to affected individuals and their caregivers. The therapies for managing dystonia vary in method and success rate.
Therapies to manage dystonia can include:
- Intramuscular botulinum toxin
- Botox is injected in spastic, dystonic muscles to help them relax for a period of time
- Artane (trihexyphenidyl), taken orally, usually divided into multiple doses each day
- Baclofen (oral or intrathecal)
- One of the main drugs used to treat dystonia, usually first taken orally and divided into several doses each day
- In the intrathecal method, an implanted baclofen pump delivers medication directly into the spinal fluid
- Deep brain stimulation
- Used increasingly more often in NBIA and has some evidence for benefit
- A stimulator sends electrical impulses to the affected brain region to help muscles relax
- It involves surgical implantation of a lead, extension and battery pack (IPG)
- The lead contains 4 electrodes and is implanted in the globus pallidus region of the brain
- The extension connects the lead to the battery pack (IPG)
- The IPG is a battery-powered neurostimulator that is placed in the abdomen (or in some cases below the clavicle)
- Physical and occupational therapy
- May or may not be indicated for those who are only mildly symptomatic
- Therapies to maintain normal joint mobility for as long as possible may be useful
The symptoms of parkinsonism can be treated with the same medications used in Parkinson’s disease. Treatment with dopamine agonist drugs (like levodopa) must be started and monitored carefully. In the beginning, the dose is increased gradually until both the patient and doctor feel symptoms are under control. While taking dopaminergic drugs, individuals must be regularly monitored for adverse neuropsychiatric effects, psychiatric symptoms and worsening of parkinsonism. There is often short-term great benefit from Parkinsons medications. However, this usually only lasts a few years and is often eventually limited by the development of dyskinesias (a common side effect that creates unwanted movement).
Even after a diagnosis has been made and the appropriate therapies have been chosen, it is recommended to continue long-term surveillance to decrease the impact of CoPAN symptoms and increase quality of life.
Long-term surveillance for CoPAN can include:
- Medication for spasticity, dystonia, and/or parkinsonism
- Monitoring of individuals receiving dopaminergic drugs for parkinsonism for:
- Adverse neuropsychiatric effects
- Psychiatric symptoms
- Worsening of parkinsonism
- Monitoring of height and weight in children
- Swallowing evaluation and regular dietary assessments
- Assure adequate nutrition
- Prevent aspiration
PROGRESSION
The symptoms of CoPAN typically progress at a slow rate. However, by the time most reported individuals with CoPAN reach their 30s, the majority can no longer walk.
The average lifespan varies for individuals with CoPAN, but due to improvements in medical care, more affected individuals are now living into middle age.
Copyright © 2014 by NBIAcure.org. All rights reserved.